All
- All
- Articles
- Calendar
- Compliance
- Newsletter
- Position Statements
- Press Release
- Regulatory
- Uncategorized

How to Work with FDA on Voluntary Recalls
By Mary Borneman, Sr. Director, Communications & Public Affairs, Standard Homeopathic Company/Hyland’s, Inc. In April 2019, U.S. Food & Drug Administration (FDA) published a...
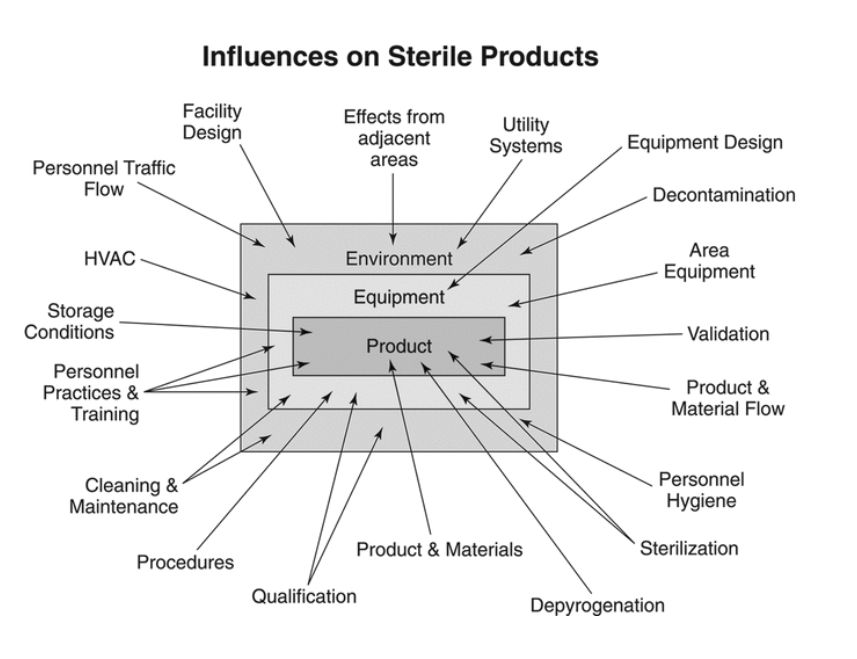
Safe Drugs, Happy Consumers: Sterility Assurance Programs
By Kristina Skowronek, Director of Quality and Regulatory Compliance, Boiron USA USP GENERAL CHAPTERS, GENERAL INFORMATION, 〈1211〉 STERILIZATION AND STERILITY ASSURANCE OF COMPENDIAL ARTICLES If...

Initiation of Voluntary Recalls: Draft Guidance for Industry and FDA Staff
By Mark Land, AAHP President Product recalls are an important tool that manufacturers and the U.S. Food and Drug Administration (FDA) use to minimize...

Overview: Membership Meeting 2019
The membership of AAHP came together on March 8 for the association’s semiannual Membership Meeting. Taking place during Natural Products Expo West in Anaheim, Calif.,...

Reduce Risk with Product Substantiation
Susan J. Hewling When bringing products to market, it is essential to build a platform that allows all information generated...

The Only Constant in Retail is Change
By Ray Petrick, Vice President of Sales, Boiron USA The retail landscape is changing—and dramatically. During the past 10 years, retail disruption has become the...

Why Pharmacovigilance Matters
By Mark Land, AAHP President Pharmacovigilance is defined as the science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or...

