All
- All
- Articles
- Calendar
- Compliance
- Newsletter
- Position Statements
- Press Release
- Regulatory
- Uncategorized

The European Pharmacopeia and How U.S. Manufacturers Can Use It
By Mark S. Phillips, Pharm. D. Jan. 26, 2017 On Jan. 1, 2017, the 9th edition of the European Pharmacopoeia (Ph. Eur.) became legally binding...
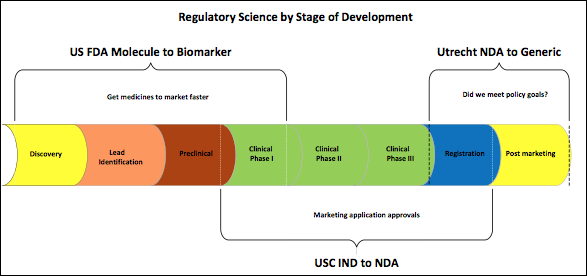
Regulatory Policy Analysis
A Tool for Evaluating the Regulatory Framework for Homeopathic Drug Products By Mark Land, AAHP President Today the combination of drug regulations, approval options, cost...

Who Regulates Your Product Labels?
Does the U.S. Food and Drug Administration (FDA) regulate homeopathic product labels? Or does the U.S. Federal Trade Commission (FTC)? Is it both? While most...

A Trump Administration and a Republican-Controlled Congress
An Assessment and Outlook of the Political and Regulatory Climate for 2017 By Pete Evich, AAHP government relations January 1, 2017 The election of Donald...

2016 Year-In-Review
As 2016 comes to a close and we think back on all the many amazing and unexpected things that have happened, we also note the...

AAHP Compliance Thru Education Webinar on FDA Inspections
The association just completed another successful Compliance Thru Education webinar: FDA Inspections of Homeopathic and Drug Manufacturing Facilities: An Agency View. This webinar, held on...

Label Comprehension Studies for Nonprescription Drug Products
The Food and Drug Administration (FDA) sometimes requires sponsors to conduct label comprehension studies that are designed to evaluate proposed nonprescription drug product labeling. A...

