All
- All
- Articles
- Calendar
- Compliance
- Newsletter
- Position Statements
- Press Release
- Regulatory
- Uncategorized

Process Validation of Homeopathic Medicines
By Pejman Parhami, Senior Director, Quality Systems, Hyland’s, Inc. FDA issued many warning letters regarding process validation in 2017 and 2018. were more prominent....
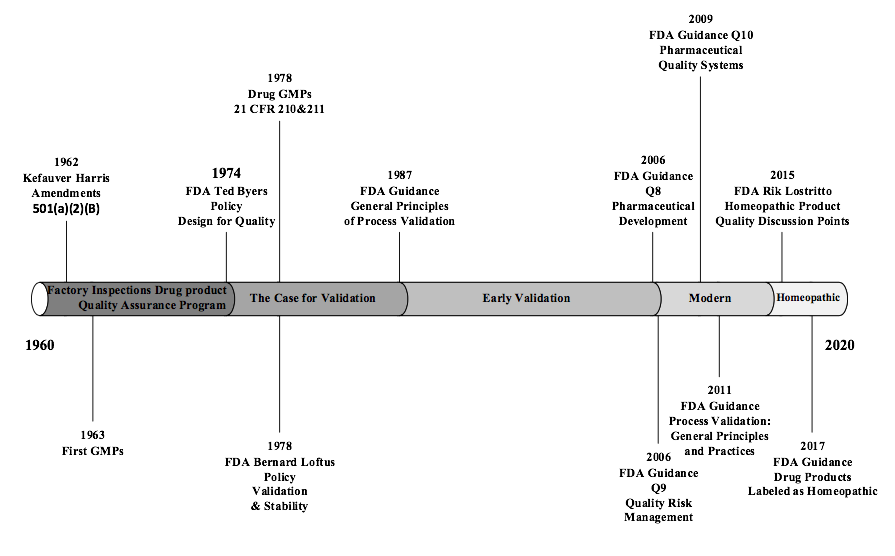
A Brief Regulatory History of Process Validation
By Mark Land, AAHP President Finished product testing is probabilistic and not dispositive of process control; that is why we need process validation. While...
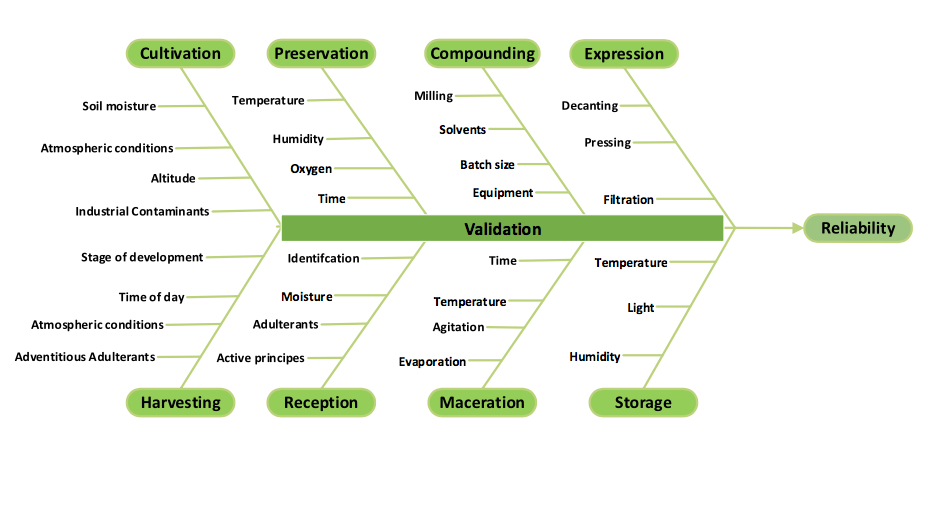
The Case for Process Validation
By Mark Land, AAHP President The Homeopathic Pharmacopoeia of the United States says: “Dosage Forms - These, like all other conditions of homeopathic production...

AAHP Mourns the Loss of Dr. Peter Fisher
AAHP, along with the global homeopathic community, is shocked and saddened to learn of the tragic and untimely death of Dr. Peter Fisher. A champion...

Staff Can Amplify Your Message on Social Media
Your employees might not all be marketers, but with the rise of social media, they can all be advocates for your company and homeopathy in...

The British Homœopathic Pharmacopœia
By Eric Foxman, AAHP Secretary A quick look at its history, purpose, and contents, as well as the potential for adulteration and misbranding violations in...

Prepare for Recalls: 5 Crucial Steps
By Ryan Gooley, Recall Consultant, Stericycle Recalls impact thousands of companies every year, and they represent serious risk for the brands impacted. Homeopathic recalls,...


